Natural radioactive background as a factor for external and internal exposure of the population
According to the Regulation on basic standards of radiation protection (RBSRP-2012) the natural radiation background is a radiation field caused by natural sources. Such natural background radiation excludes all man made or technologically enhanced radiation pollution.
“Natural source” is a source of ionising radiation, existing under natural conditions – cosmic radiation and naturally spread radionuclides in the environment, in foods and in human body.
Life on Earth has appeared and exists in the environment of natural ionising radiation. According to contemporary science the age of the Universe is estimated to be about 15 billion years and the Earth, as one of the planets in the Solar system, emerged 4.5 billion years. Solar light and heat, due to which life on Earth exists, are obtained as a result of nuclear reactions in the Sun. Nuclear reactions occur also in galactic space.
During these nuclear reactions there appear powerful flows of highly energetic ionised particles, which form the primary cosmic rays. They consist of two components, which significantly differ by their origin as well as by the composition, intensity and energy:
- galactic cosmic rays;
- solar cosmic rays.
The intensity of the galactic cosmic rays, which reaches the Earth, is constant and its influence is isotropic (uniform in all directions towards the Earth). They are composed of protons (atomic nuclei of Hydrogen) – about 90 %, α-particles (atomic nuclei of Helium, 10 %), and about 1 % photons, neutrons, electrons and atomic nuclei of some light elements (Lithium, Beryllium, Carbon, Oxygen, Nitrogen) which originate in the depth of the Universe as a result of emission and evaporation of matter during explosion and creation of new stars.
The intensity of the solar cosmic rays is not constant and varies depending on the solar activity. The cycle of changes in solar activity is 11 years.
When the primary cosmic rays reach the atmosphere of the Earth, the high energy ionising particles interact with the atoms and molecules of the air-creating multitude of electrically charged and neutral ionising particles. They, in turn, create “avalanches” of new ionising particles in the atmosphere and thus create secondary cosmic rays, which consist of photons (γ-quanta), electrons, neutrons and other elementary particles. The intensity of the secondary cosmic rays has a maximum at an altitude of 20-25 km. Above the surface of the Earth. With the decrease in altitude its intensity decreases and reaches its minimum at sea level.
The intensity of the cosmic rays depends on the altitude as well as the place (geographical latitude) of a given region of the Earth. The intensity of the cosmic rays at the poles of the Earth is about 10 % higher compared to the equatorial regions.
The atmosphere that envelopes the Earth and spins together with it extends to an altitude of about 1 000 km. The atmosphere may be described as a “shield” which covers the Earth and absorbs the greater part of the cosmic rays, thus slowing or stopping the powerful flow of high energy particles coming from Space. Owing to this “anti radiation shield” the ionising radiation, which falls on Earth, is significantly decreased. With the increase of the altitude the thickness of the protective atmosphere layer decreases and that is why the exposure to cosmic rays in high mountainous areas is greater compared to that at sea level, where the protective layer is thickest and more dense. For example, at Mount Everest, the highest point on Earth (8848 m.), the influence of the cosmic rays has 20 times greater intensity than at sea level.
Earth has one more natural shield, which mitigates the impact of the cosmic rays on its inhabitants – this is the Earth’s magnetic field. Earth resembles a giant magnet whose magnetic field deflects part of the electrically charged particles coming from Space forcing them to circumvent our planet. The other part of the charged cosmic particles under the influence of the magnetic field of the Earth are deflected (whirled) in the equator area and concentrate in the regions of the geographical poles following the configuration and the direction of the magnetic field lines surrounding the Earth. That is why the intensity of the cosmic rays in the equatorial areas is minimal while in the areas of the North and South poles it is maximal. Due to the existence of the Earth’s magnetic field the intensity of the cosmic rays changes depending on the latitude. For example, at about 50 degrees northern latitude (London, Moscow, Tokyo, New York) the exposure from cosmic rays is about 30 – 40 % higher compared to areas within 15 degrees latitude of the equator.
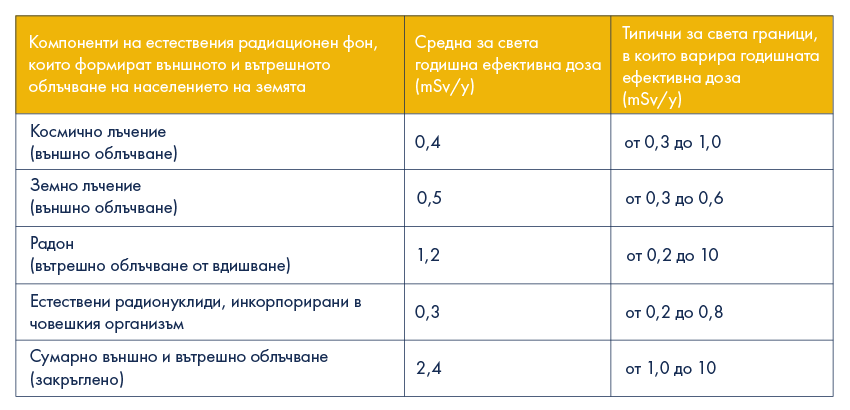
Cosmic radiation is a natural phenomenon and exposure to it cannot be avoided or be managed in any way by men. The typical value of the average annual individual dose, which is received by external exposure from cosmic radiation, for middle Earth latitudes, including Bulgaria, is 0.4 mSv/y (0.4 millisievert per year).
The Earth’s crust (soils, rocks, minerals) and the living environment (air, water, flora, fauna) contain various natural radionuclides from cosmic and terrestrial origin. Natural radioactive elements are contained also in all foods and in the human organism.
Natural radionuclides of terrestrial origin may be grouped in the following way:
- radionuclides from the existing in nature three natural radioactive chains of decay: uranium-radium chain with parent radionuclide Uranium-238 (half-life 4,5 billion years), thorium chain with parent radionuclide Thorium-232 (half-life 14 billion years) and actinium chain with parent radionuclide Uranium-235 (half-life 700 million years). All these chains contain 47 different radionuclides.
- radionuclides from the middle part of the periodic table of elements – 12 in total, all with half-life more than 10 billion years, from which the main are Potassium-40, Rubidium-87, Calcium-48.
These radionuclides, which are formed as a result of the primary processes during the formation of the Earth as a planet, are called “primary radionuclides”. The highest content of natural radionuclides is found in volcanic rocks (for example granite). The content of natural radionuclides is also high in sediments such as clay, phosphates and other natural minerals.
Due to the extremely long half-lives, commensurable with the age of the Earth, the parent radionuclides and the progenies of the three natural radioactive families, as well as the rest of the natural radionuclides (Potassium-40 etc.) continue to exist in the crust of the Earth up to the present.
The presence of natural radionuclides on the surface of the Earth leads to external exposure. The dose rate of the earth’s exposure is directly proportional to the amount (concentration) of natural radionuclides in the soil. The concentration of radionuclides in the soil depends on the radioactivity of the substances from which the soil is composed and on the processes of sorption, deposition and washout of radionuclides upon the influence of natural waters on it.
The natural γ-background on the surface of the Earth is created mainly from radionuclides which are contained in the upper soil layer with thickness of about 30 cm. Due to the shielding effect of the soil the γ radiation from natural radionuclides lying at a depth greater than 30 cm. significantly diminishes and does not play a significant role for the formation of the γ – background.
35 % of the external exposure as a result of the natural γ–background from the Earth’s surface is due to Potassium-40, 25 % – of the γ emitting radionuclides from the Uranium-238 chain (mainly Lead-214 and Bismuth-214) and 40 % of the γ emitting radionuclides from the Thorium 232 chain (mainly Thallium 208 and Actinium-228). The investigations in many countries show that it is completely normal that the natural γ–background, caused by the Earth’s radiation, to differ up to 6 times for different regions of the Earth. Standard practice is to measure natural γ–background at a height of 1 m above the ground.
The population living in mountainous places and regions rich in granites, minerals and soil with high concentration of natural radionuclides is subjected to higher external exposure. The γ–background in such places is higher.
When the exposure of the population to natural radionuclides is evaluated it should be taken into account that for a significant part of his time a person stays in the premises where he lives and works. In principle, the walls, floors and roofs of the buildings shield (weaken) the natural γ–background. On the other hand, however, construction materials (bricks, concrete, plasters) from which buildings and homes are made contain natural radionuclides with various concentrations. This leads to the exposure of the people living in the buildings. The γ–background inside buildings constructed from bricks, concrete and granite, as a rule, is higher than that in wooden constructions. The results of investigations conducted in the world show that the γ–background inside buildings is, on the average, 20 % higher than in the open air. This depends on the content of natural radionuclides in the construction materials used and from the local gamma background in the open.
To assess of the average annual effective dose for the population on Earth, the exposure from natural radionuclides indoor and outdoor should be considered separately. It is estimated that modern man spends approx. 80% of his time in buildings (19 hours per day), and 20% outdoor (5 hours per day), which means that the natural indoor exposure of a person dominates the outdoor one.
It is established that the gamma-radiation, caused by the natural radionuclides in the ground surface layer and in building materials, causes external exposure, which, averaged over the world, is estimated to be 0.5 mSv per year (0.5mSv/y). For 95% of the Earth’s population the average individual annual effective dose from the natural (coming from the ground) gamma-radiation varies between 0.3mSv/y and 0.6mSv/y.
The γ-background (coming from the ground) varies during rain and snowfall. The blanket of snow decreases the γ-background with 10%, up to 20% in some areas, because of the shielding effect. Rains bring down to the ground surface natural radionuclides (radioactive aerosols), contained in the near the surface ground air (water washes down the dust particles in the air) and as a result of this, the γ-background could increase in some areas by 20% to 50% (especially in heavy rains or at the initial hours of the rain). The influence of the meteorological factors should always be taken into account, when the γ-background of a given area is assessed, because the short duration sample values could vary up to 50% for some time interval (hours or days) from actual long term averages.
And in conclusion, in the world average effective dose from external exposure for the population, due to cosmic rays (0.4mSv/y) and the ground radiation (0.5 mSv/y), amounts to 0.9 mSv/y.
In addition to the external exposure, the natural radiation background causes internal exposure when certain natural radionuclides get into human body by inhalation and swallowing.
The main radionuclides from the Uranium-Radium and Thorium chains, which cause internal exposure to humans, are Uranium-238, Radium-226 and Thorium-232. The radionuclides with terrestrial origin get into the human body through inhalation of dust particles, lifted from the ground and by swallowing of food and water. Excluding Polonium, which accumulates in the soft tissues, all other natural alpha-nuclides accumulate mainly in human bones. The internal exposure from Thorium-232 is about 3 times less than that from Uranium-238. From all known 13 Radium radioisotopes, the main contribution for the internal exposure comes from Radium-226, which enters the human body mainly through food.
Another main source for the natural internal exposure is Potassium-40, which enters the human body mainly through food. The natural Potassium contains 2 stable isotopes: Potassium-39 (93.08%) and Potassium-41 (6.91%) and one unstable (radioactive) isotope – Potassium-40 (0.01%) with half-life 1.3 billion years. Everywhere in nature this percentage is the same for the three Potassium isotopes. Every living organisms in nature contains Potassium, which is an essential bio-element. Living organisms, assimilating Potassium, don’t change its nuclide content.
The content of Potassium-40 in food varies in wide ranges – from 20 up to 600 Bg/kg. In the human body, depending on age, Potassium concentration varies between 1.0 and 2.5 g/kg. Potassium is concentrated in human muscles. In young people Potassium concentration, Potassium-40 respectively, is approx. 2 times higher than in adults. The average specific activity of Potassium-40 in human body is 60 Bq/kg.
The average annual effective dose, which the Earth’s population receives from internal exposure to natural earth radionuclides (mainly Uranium-238, radium-226, Thorium-232 and Potassium-40), contained in the human body, is estimated to be 0.3mSv/y.
Cosmogeneous radionuclides are formed mainly as a result of the interaction between the cosmic rays and the atomic nuclei in the air and to a lesser degree by the interaction with atomic nuclei, contained in the Earth’s crust. There are about 20 cosmogeneous radionuclides known, the main being Tritium, Carbon-14, Berilium-7, Sodium-22, Phosphorus-32, Sulphur-35 and Chlorine-36.
To estimate of the internal exposure from cosmogeneous radionuclides, the radioactive impact of Carbon-14 and Tritium should be taken into account (for Carbon-14 the annual effective dose from internal exposure is about 0.001 mSv/y, and for Tritium is much less). The internal exposure caused by radionuclides with cosmogeneous origin, is negligible, compared with the internal exposure due to radionuclides with terrestrial origin.
The major contribution to the internal exposure of the Earth’s population has the natural radioactive element Radon (Rn-222) with atomic number 86 in the Mendeleev periodic table. The average annual effective dose, obtained by a person as a result of radon inhalation and its short-lived progenies in air, is estimated to be 1,2 mSv/y. Hence the natural internal exposure, due to Radon and its progenies (1.2mSv/y) is 4 times greater than the internal exposure, due to the natural radionuclides with cosmogeneous and terrestrial origin. (0.3mSv/y).
Radon (Rn-222) is a radioactive inert (noble) gas, heavier than the air. It is a result of alpha-decay of the atomic nuclei of Radium (Ra-226), which is a progeny in Uranium-Radium chain, with the parent radionuclide Uranium 238. After the radioactive decay of the gaseous Radon-222 (3.8 days half-life) a whole chain of new products in solid aggregate state are formed, namely Polonium-218, Polonium-214, Bismuth-214 and Lead-214 (short-living Radon progenies with half life under 30 minutes). These Radon progenies stick to the existing microscopic dust particles in the air (aerosols) and when inhaled, they are retained in the human respiratory system.
The internal exposure is precisely a result of inhalation of short-living progenies, not the inhalation of the radon itself, which is an inert (noble) gas. From the radiation protection point of view all noble gases are a source for external exposure, because by nature they are chemically inert gases and do not participate in biochemical reactions with substances contained in human tissues and organs. The noble gases are also easily exhaled from the lungs. As a result of many scientific investigations it was established, that 98% of the human internal exposure is due to inhaling of radon progenies, which influence the bronchial epithelium in the lungs, and only 2% of the exposure is due to Radon-222 itself.
Natural Uranium is spread in the whole Earth’s crust and its content is estimated to be approx. 3.10-4 % (average about 3 g per ton of soil). It follows that radon as a member of the Uranium-Radium chain, is also distributed everywhere on the Earth – in soils, rocks, minerals, waters, air. A proportion of radon generated in the natural chain Uranium-Radium-Radon, gets into the air from the ground (soils, rocks and water). The radon distribution by air into the environment by diffusion and convection depends on some natural and climate factors – atmospheric pressure, air temperature, rainfalls, wind, topography and other characteristics.
The radon concentration in the air decreases with altitude. In addition, the radon concentration in air changes with the seasons – the highest values are registered during summer months. During twenty-four-hour period radon concentration also varies (the maximum is during the night, minimum – during the day).
Typical average radon concentration in open air near the ground is 10 Bq/m3. As a rule, the radon concentration in buildings is higher and depending on the building materials. The typical radon concentration in buildings is 40 Bq/m3.
Thus, the annual effective dose from internal exposure for the population, due to Radon (1.2 mSv/y) and to other radionuclides with cosmogeneous and terrestrial origin (0.3 mSv/y), is totally equal to 1.5 mSv/y.
Hence the world average for annual effective dose from both external and internal exposure, due to the natural radioactive background is 2.4 mSv/y. 50% of this dose (1.2 mSv/y) is due to radon and its short-lived progenies, contained in the air, 37.5% (0.9mSv/y) – due to external terrestrial and cosmic rays, and 12.5% (0.3mSv/y) – due to radionuclides with terrestrial origin, incorporated in the human body.
Table № 1
In Table 2 are shown generalized data for the exposure of the European population, due to natural radiation background.


 Български
Български


